Used previously for dengue virus studies, this cynomolgus macaque model will serve to better understand synergy between these viruses
In an effort to help identify a vaccine for the Zika virus (ZIKV), Southern Research is actively running experiments to study the course of ZIKV infection in non-human primates, while developing animal models for the evaluation of candidate vaccines and drug therapies. Cynomolgus macaques were chosen for these studies as they have a relatively simple HLA phenotype that allows for a reproducible immunological response. Additionally, the Cynomolgus macaque was previously established at Southern Research as a relevant animal model for dengue viruses (DENV), and has been successfully used to evaluate new candidate DENV vaccines. This model will allow the study of viral synergy to better understand the impact of cross-reactive but non-neutralizing antibodies from DENV infection/vaccination on ZIKV pathogenesis.
In this study, a total of six (6) male and female ZIKV and DENV seronegative Cynomolgus macaques were challenged with ZIKV strain isolates PRVABC59, FSS13025 or IBH 30656 subcutaneously (n=2 per strain) with a target delivery dose of approximately 1×104 or 5×105 PFU per monkey. A description of the viruses used are shown in Table 1.
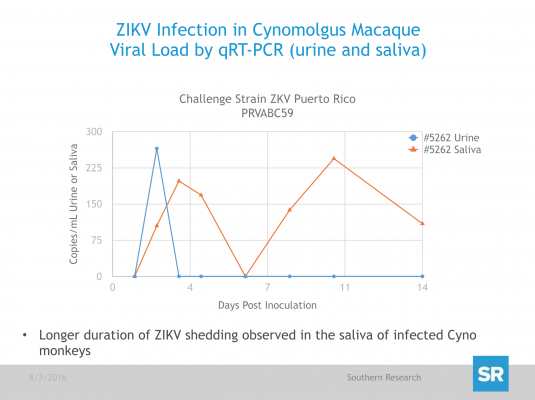
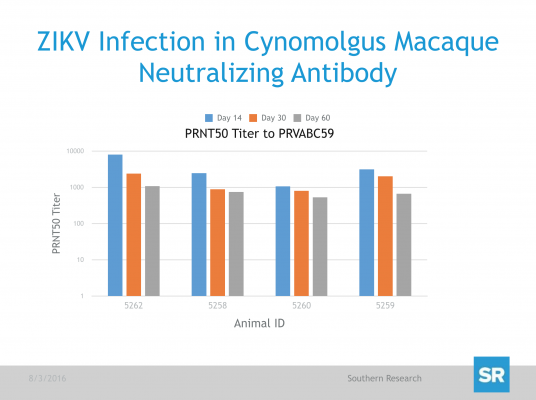
After ZIKV inoculation, serum and tissue samples were collected at predetermined time points and are being analyzed by quantitative real-time polymerase chain reaction (qRT-PCR) for the detection of viral load. Additionally, Day 14, 30 and 60 serum samples will be analyzed by Plaque Reduction Neutralization Test (PRNT) for the enumeration of ZIKV neutralizing antibody (Nab) as a potential surrogate of protective immunity.
Table 1. Zika virus strains evaluated in a non-human primate model of infection.
| Zika Virus Isolate | Source | Year of Isolation | Geographical Origin |
| PRVABC59 | CDC | 2015 | Puerto Rico (AM) |
| FSS13025 | UTMB arbovirus Reference collection | 2010 | Cambodia (AS) |
| IbH 30656 | BEI Resources | 1968 | Nigeria, West Africa (AF) |
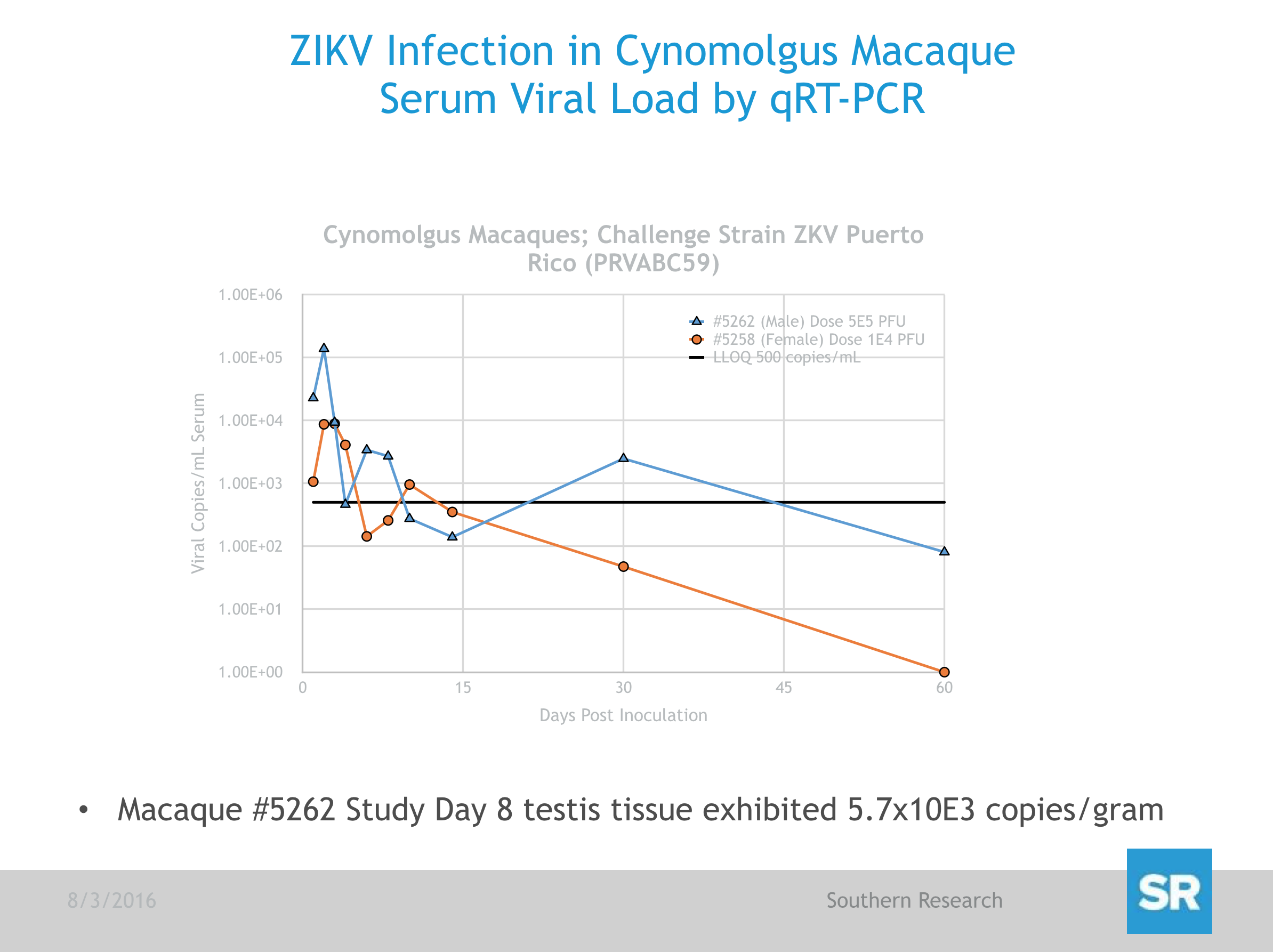
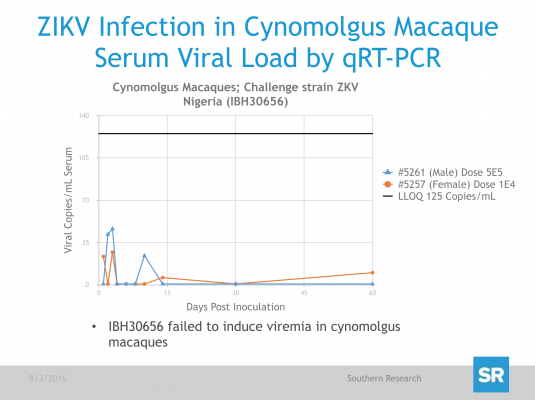
Preliminary results of this study demonstrated no significant changes in body weight, body temperature or clinical signs in animals challenged with ZIKV, as was expected from studies reported by other groups in Rhesus macaques and consistent with DENV infection of this model. However, ZIKV RNA was detected by qRT-PCR in the serum of primates challenged with the Puerto Rican strain of ZIKV (PRVABC59) as soon as 1 day post-challenged and peaked after Days 2 or 3 but remained detectable out to 14 days (Fig. 1). In contrast, ZIKV RNA concentrations in serum from those animals challenged with the Cambodian Strain (FSS13025) fell below the level of detection on Days 8 or 10 but increased again by Day 14 in at least one of the 2 animals. SR is continuing to assess viral load in serum and tissues from all primates on study, and data will be updated as soon as it becomes available.
Preliminary data for Cynomolgus Macaque model (updated: 8/03/16)
Click the images below to enlarge.